Developed for the life science and pharmaceutical industries – FDA and GxP ready
The regulations in the pharmaceutical industry are strict. Octoplant from AMDT helps you comply with them automatically. Everything is documented, tracked and saved, allowing you to handle audits and self-checks without stress.
With octoplant you get:
- Automated version management and backup of all systems throughout the production chain.
- Full traceability and documentation in accordance with FDA (21 CFR Part 11) and GxP
- Effective management of changes in production systems
- Early detection of errors and improved quality assurance
- Facilitated compliance with legal requirements and smoother audits
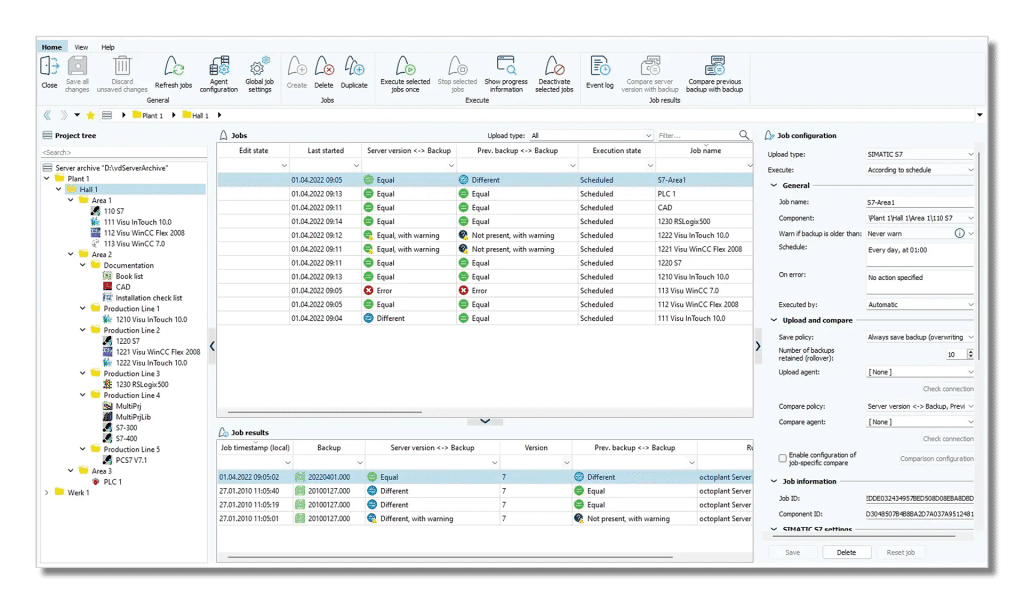
Version control that makes a difference
Action and results :
With Octoplant you get full control over who changed what, when, where and why . Whether it’s PLC programs, HMI files or robot control, everything is documented automatically.
Result :
Total transparency, better quality and faster recovery.
With Octoplant, you automate documentation processes, strictly follow FDA and GxP regulations, and ensure traceability throughout the production chain.
Result :
Fewer errors, better efficiency, and more time to focus on safe and effective medicines.
Try free for 30 days
- No installation required.
- Automatically terminates after 30 days.
- Extension of the trial period is possible at any time.
Minimise downtime – restore production in minutes
In the event of unexpected errors or interruptions, every minute can cost money and can even jeopardize product safety. With octoplant’s automatic backups, you can restore to the correct system configurations in just a few minutes.
- Clear overview of latest and correct versions
- Pattern-based or custom recovery
- Alarms in case of deviations in parameters or settings
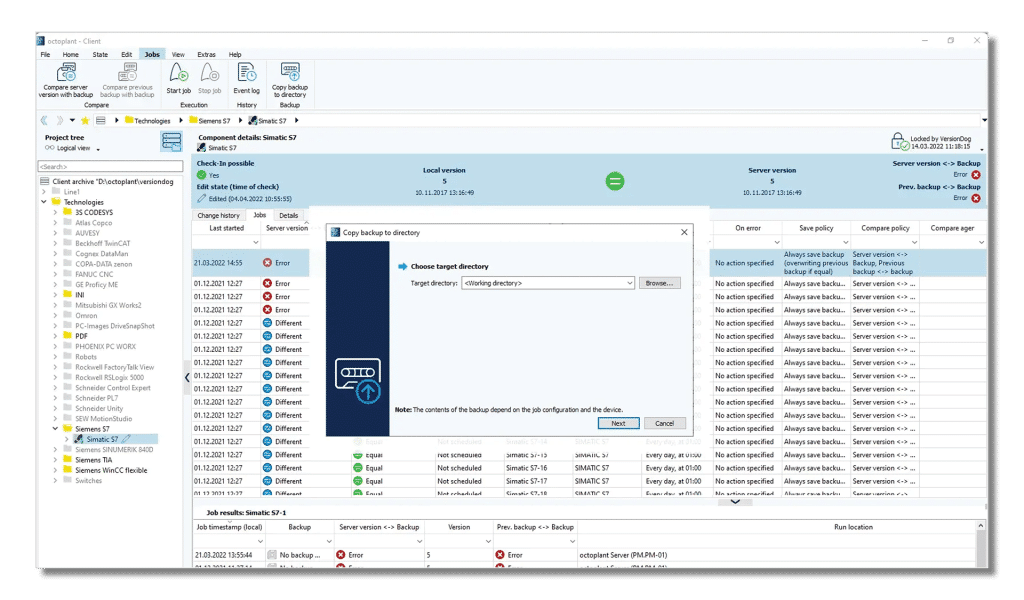

Audit trails and risk reports – ready for the next inspection
Avoid last-minute audit panic. With Octoplant, audit trails are just a click away – detailed, verifiable and fully traceable. This makes it easy to show complete documentation and ensure you meet all regulatory requirements.
- Get risk reports with detailed analysis of assets
- Document even small changes to projects
- Generate audit trails with one click
- Establish industry-standard approval flows

